Generic drug substitution is the practice of dispensing a different brand or unbranded medication for the one prescribed by a physician. For pharmacists, this isn't just about swapping pills to save money. It’s a high-stakes legal and clinical decision that requires precise documentation. Get it wrong, and you risk patient safety issues, insurance denials, or even losing your license. Get it right, and you help keep healthcare costs down while ensuring patients get effective treatment.
Why Documentation Matters More Than You Think
You might think that if two drugs are "bioequivalent," they’re interchangeable without question. That’s a dangerous assumption. While the FDA ensures generics meet strict standards-identical active ingredients, dosage form, strength, and route of administration-they don’t regulate the actual act of substitution at the pharmacy counter. That falls to state laws. The stakes are real. Inadequate documentation led to a documented case in 2021 where a patient was hospitalized due to unmonitored INR fluctuations after a warfarin substitution wasn’t properly recorded. Warfarin is a Narrow Therapeutic Index (NTI) drug, meaning small changes in blood levels can be life-threatening. Proper documentation didn’t just protect the pharmacist; it saved the patient’s life. Conversely, comprehensive documentation has proven benefits. A 2022 study showed that pharmacies with robust substitution records reduced medication errors by 17.3% over six months. When you document correctly, you create a clear trail for prescribers, insurers, and other healthcare providers.The Core Elements: What Must Be Documented?
Across the United States, requirements vary, but there’s a common baseline. According to surveys from the National Association of Boards of Pharmacy, 48 states require pharmacists to record specific details when substituting a generic for a brand-name drug. Here’s what typically needs to go into the prescription record:- Brand name prescribed: The exact drug name written by the prescriber.
- Generic name dispensed: The non-proprietary name of the medication given to the patient.
- Manufacturer name: Who made the generic version? This matters because manufacturing processes can differ slightly between companies.
- Lot number: Essential for tracking recalls or quality issues.
- Expiration date: Ensures the patient receives a potent, safe product.
Navigating State Laws: The Patchwork Quilt
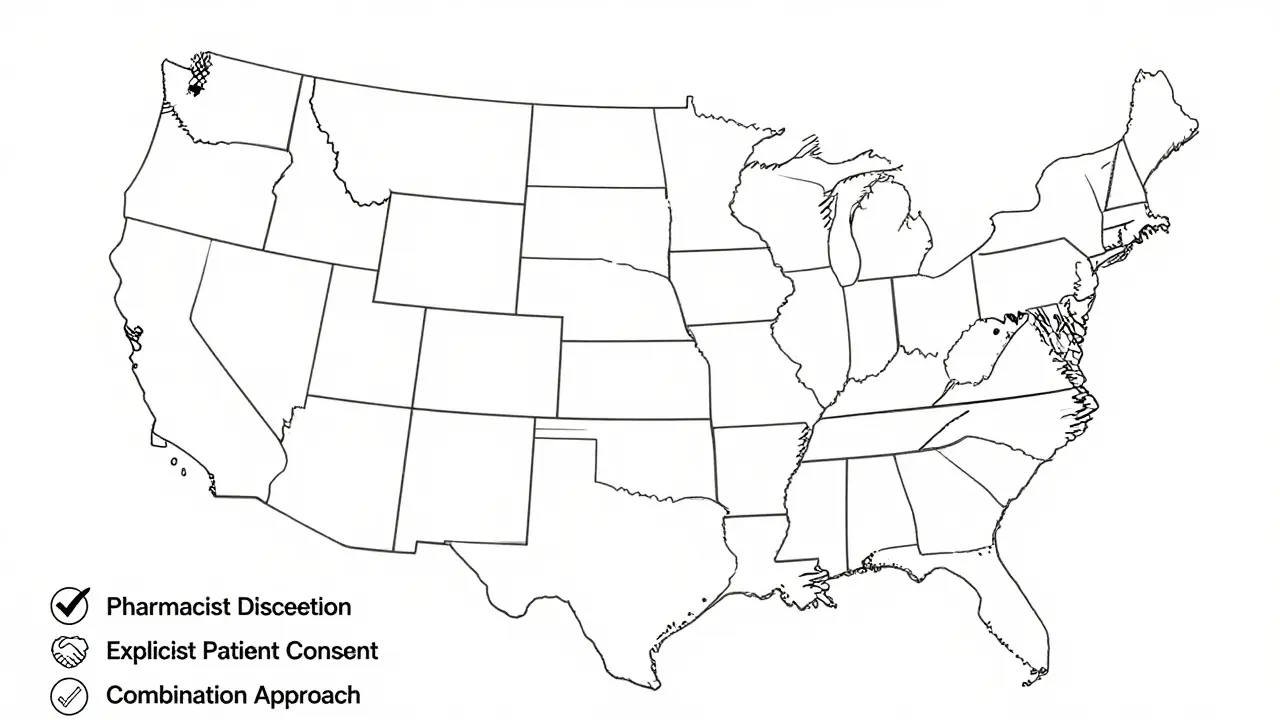
This is where things get tricky. There is no single federal rule for how pharmacists must document substitutions. Instead, you’re dealing with a patchwork of 50 different state regulations.| Approach Type | Number of States | Key Requirement | Patient Consent Needed? |
|---|---|---|---|
| Pharmacist Discretion | 27 | Document substitution in pharmacy records | No |
| Explicit Patient Consent | 14 | Record patient agreement before substitution | Yes |
| Combination Approach | 9 | Varies by drug type or situation | Sometimes |
Special Handling: Narrow Therapeutic Index (NTI) Drugs
Not all generics are created equal in the eyes of regulators. NTI drugs require extra caution because small differences in bioavailability can lead to serious adverse effects. Examples include warfarin, levothyroxine, and phenytoin. While the FDA hasn’t formally designated NTI drugs in its Orange Book, 17 states have established specific documentation protocols for them. These usually involve:- Additional physician notification before substitution.
- Explicit patient consent beyond standard procedures.
- Enhanced monitoring plans documented in the patient’s record.
Electronic Health Records: Friend or Foe?
Most pharmacies now use electronic systems to manage prescriptions. However, these systems aren’t always set up to handle state-specific documentation requirements out of the box. Epic Systems reported that 32% of pharmacies using their platform needed custom configuration to meet state laws. This means you might need to work with your IT team or software provider to ensure your system captures all necessary fields. Look for features like:- Automated prompts for NTI drug substitutions.
- Fields for manufacturer lot numbers and expiration dates.
- Integration with prescriber portals for real-time updates.

Best Practices for Smooth Documentation
Mastering state-specific documentation takes time. New pharmacists typically spend 4-6 weeks learning the ropes. To speed up the process and reduce errors, consider these best practices:- Know Your State’s Law: Use resources like the National Community Pharmacists Association’s online tool, which cross-references state laws quarterly.
- Standardize Your Workflow: Create checklists for common scenarios, especially for NTI drugs.
- Train Your Staff: Ensure everyone understands why documentation matters and how to do it correctly.
- Review Regularly: Laws change. Set reminders to review updates annually or biannually.
- Communicate with Prescribers: If a substitution raises concerns, call the doctor. Document the conversation.
Looking Ahead: Standardization and Technology
The future looks promising for simplifying documentation. The Model State Pharmacy Act, updated in 2022, recommends uniform requirements across states. Twelve states have already adopted these guidelines as of 2023. Technology is also playing a role. Blockchain pilots have shown a 22% reduction in documentation errors by creating immutable, shared records of substitutions. Meanwhile, the WMA now recommends that electronic health record systems include standardized fields for documenting substitution decisions and patient outcomes. With potential national guidelines expected in Q2 2024, we may soon see less fragmentation. Until then, stay vigilant, stay informed, and document everything.Do I need patient consent to substitute a generic drug?
It depends on your state. In 14 states, explicit patient consent is required. In 27 states, pharmacists can substitute at their discretion without direct consent, though they must still document the action. Nine states use a mix of both approaches. Always check your local regulations.
What information must I document when substituting a generic?
Typically, you need to record the brand name prescribed, generic name dispensed, manufacturer, lot number, and expiration date. Some states require additional details, especially for NTI drugs or when patient consent is involved.
How do I handle Narrow Therapeutic Index (NTI) drugs?
NTI drugs require extra care. Seventeen states mandate additional physician notification and patient consent. Always verify your state’s specific rules and consider implementing enhanced monitoring plans documented in the patient’s record.
Is my pharmacy software automatically compliant with state laws?
Not necessarily. About 32% of pharmacies using major EHR systems like Epic need custom configurations to meet state documentation requirements. Work with your IT provider to ensure all necessary fields are captured and accessible.
What happens if I fail to document a substitution properly?
Poor documentation can lead to insurance claim denials, patient safety issues, and legal liability. In extreme cases, it could result in disciplinary action against your pharmacy license. Accurate records protect both you and your patients.
Are there any upcoming changes to documentation requirements?
Yes. The ASPE is developing national guidelines expected in Q2 2024, which could reduce variation among states. Additionally, twelve states have adopted the Model State Pharmacy Act’s recommendation for uniform documentation standards.