Adverse Event Classification Assistant
This tool helps distinguish between "Severity" (how bad it feels) and "Seriousness" (medical/regulatory consequence). Review your situation to see if it fits the FDA's definition of a Serious Adverse Event.
No intervention needed
Minimal treatment
Interferes with daily function
When you read your medication guide or sign up for a clinical trial, the term Serious Adverse Event often appears in bold print. It sounds alarming, and for good reason, but the medical meaning is more specific than most people realize. Many patients confuse "serious" with "severe," leading to unnecessary panic when reviewing trial results or experiencing side effects. Understanding how the U.S. Food and Drug Administration classifies these events gives you a clearer picture of your health risks and helps you know exactly when to seek immediate care.
What Defines a Serious Adverse Event?
The FDA defines a Serious Adverse Event through strict regulatory guidelines found in the Code of Federal Regulations. It is not just about how bad a symptom feels; it is about the specific outcome of the medical occurrence. An event qualifies as serious if it meets at least one of five critical criteria established to protect public health. These criteria were formalized to ensure that significant safety issues are detected early, allowing regulators to update warnings or halt unsafe practices quickly.
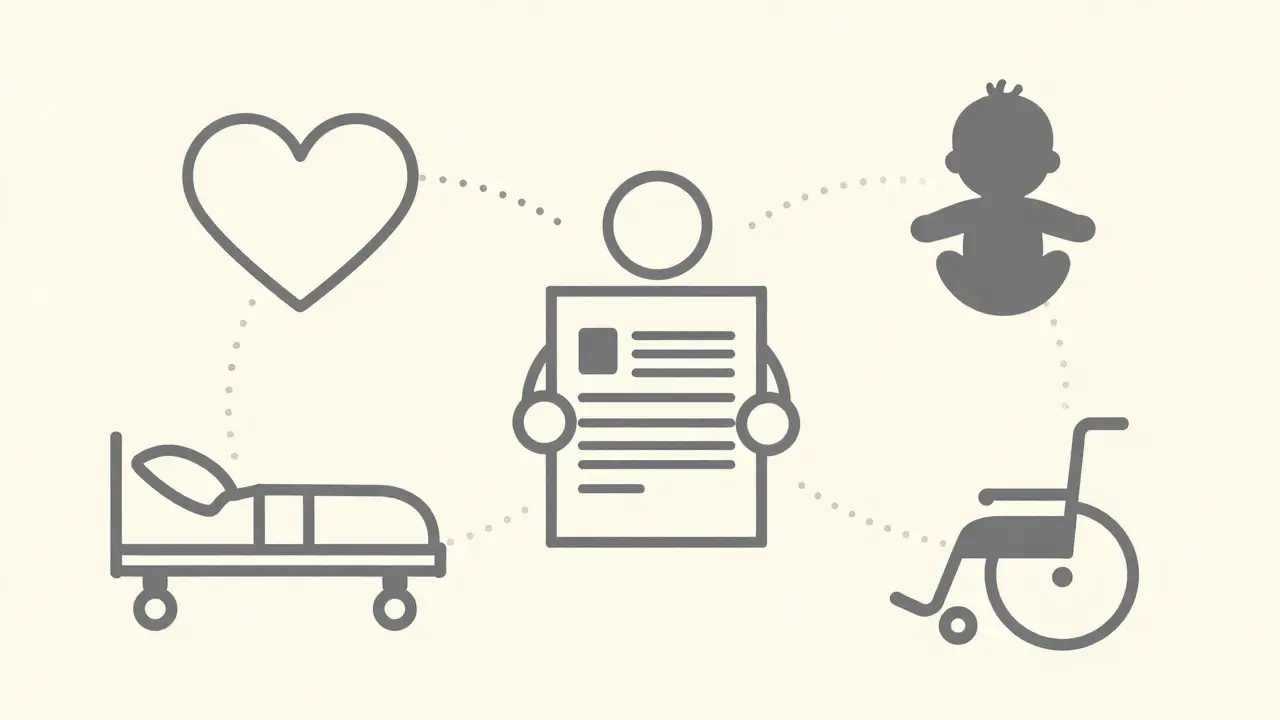
A Serious Adverse Event (SAE) is any undesirable medical occurrence associated with drug use that results in death, life-threatening situations, hospitalization, permanent disability, or congenital anomalies. This classification system ensures timely detection of risks that impact patient welfare.First, there is death. This includes any case where a participant passes away during treatment, even if the cause isn't immediately linked to the drug. Second, life-threatening status means the patient was at immediate risk of dying when the event occurred. Third, hospitalization covers both initial admission and prolonged stays extending beyond twenty-four hours. Fourth, disability implies a substantial disruption of normal life functions, preventing daily activities. Finally, congenital anomaly or birth defect refers to abnormalities present at birth resulting from exposure.
Beyond these five outcomes, there is a category called Important Medical Events. These might not always require hospitalization initially, but without intervention, they could lead to serious harm. For example, acute asthma attacks or rapid drops in blood sugar might fit here. Capturing these ensures safety signals aren't missed until damage occurs.
Serious Versus Severe: Why the Difference Matters
One of the biggest sources of confusion lies in the difference between seriousness and severity. A common misconception is that a painful or difficult side effect automatically counts as serious. However, the FDA distinguishes these two concepts clearly. Severity describes the intensity of the symptom, while seriousness describes the consequence of the outcome.
You will often see severity graded on a scale used by organizations like the National Cancer Institute. Grade 1 is mild and requires no intervention. Grade 2 is moderate and needs minimal treatment. Grade 3 is severe, requiring invasive intervention, but it might not meet the criteria for a serious adverse event if it doesn't result in hospitalization or other specific outcomes. Conversely, an event can be serious without being severe. For instance, mild dehydration that requires hospitalization is serious because of the admission, even if the physical sensation was manageable.
| Characteristic | Serious | Severe |
|---|---|---|
| Definition | Outcome-based (Death, Hospitalization) | Intensity-based (Mild, Moderate, Severe) |
| FDA Reporting | Must be reported within 15 days | Not necessarily required for regulatory reporting |
| Impact on Label | Cause warning label changes | Influences dosage recommendations |
| Patient Action | Seek immediate medical attention | May be monitored at home |

Recognizing the Criteria in Your Documentation
Patients participating in clinical trials often see lists of potential risks in consent forms. These documents must explicitly state how adverse events are monitored. You should look for sections titled "Risks and Discomforts" or "Adverse Events." They often include a glossary defining serious adverse events using plain language. When reading these, pay close attention to the incidence rates. If a document states "Serious infections occurred in 2.3% of patients," that percentage reflects the frequency of those specific five outcomes listed earlier.
Understanding this distinction empowers you. If you experience a symptom that feels uncomfortable but does not require a trip to the hospital, it may not technically be a serious event in the regulatory sense, even if it bothers you. However, if that symptom leads to an overnight stay in the ER, it instantly escalates to a serious classification regardless of how intense the pain felt beforehand. This nuance is why many patients report anxiety when seeing high-grade adverse events in public trial databases-they assume "Grade 3" always means dangerous, which is not always true.
How to Report Side Effects
Even if you are not in a clinical trial, you have the right and ability to report safety concerns directly to the agency. The FDA operates the MedWatch program, which accepts voluntary reports from patients and healthcare providers. You can access Form 3500B online to submit details about unexpected reactions to prescription drugs, biologics, or medical devices. In 2022 alone, thousands of patient-submitted reports helped identify new safety patterns that weren't obvious during initial testing.
Safety Reporting Program MedWatch is the FDA's voluntary reporting program for adverse events related to human health products. It enables patients to contribute data that supports ongoing drug safety monitoring.Reporting isn't just about protecting yourself; it contributes to the larger dataset used for post-marketing surveillance. The FDA uses this information to issue safety alerts or update labeling instructions. While underreporting remains a challenge, estimated at capturing only 1% to 10% of actual events, every submission adds value. When reporting, include details about the product name, dosage, and the timeline of symptoms. Be honest about whether other factors could have contributed, as accurate data improves the quality of safety analysis.
Navigating Patient Education and Support
Trial sponsors are required to provide education during the informed consent process. This discussion usually takes fifteen to twenty minutes and should cover exactly what happens if an adverse event occurs. During this conversation, ask specific questions. Ask how the team defines hospitalization or life-threatening scenarios in the context of your condition. If you are confused about whether a symptom requires calling the study doctor, clarify the threshold before you leave the office.
Many patients rely on community support groups for clarification after reading technical documents. Online forums often feature discussions where experienced participants share how they interpreted SAE warnings in their own treatment journeys. While peer advice is valuable, always verify information with your clinical team. Regulatory standards evolve, and guidance from 2023 onward emphasizes plain language summaries in registries. As we move into 2026, digital portals aim to make these safety summaries more accessible, reducing the gap between complex regulatory terms and patient understanding.
Frequently Asked Questions
Is a severe headache always considered a serious adverse event?
Not necessarily. A headache is classified as serious only if it leads to hospitalization, causes permanent disability, or is life-threatening. A severe pain that you treat with rest or medication at home is likely just severe in severity, not serious in outcome.
Why do clinical trial forms list so many scary sounding side effects?
Regulations require full transparency on all potential risks. Many listed events are rare theoretical possibilities rather than common outcomes. Reading the incidence rates next to the event names helps put the risk in perspective relative to how frequently they actually occur.
Can I report a side effect if I am not in a clinical trial?
Yes. You can use the FDA MedWatch form to report adverse experiences with approved medications or devices available on the market. Patient reports are vital for detecting safety issues that appeared after the drug was released to the public.
Does mild allergic reaction count as a serious event?
Generally, no. Mild hives or itching are not serious unless they progress to anaphylaxis or respiratory distress. Once an allergic reaction involves breathing difficulty requiring emergency care, it meets the life-threatening or hospitalization criteria and becomes serious.
Who determines if an event is related to my medication?
Your healthcare provider assesses the causality. For serious events, companies must report to the FDA regardless of whether the link is certain or suspected. The investigation continues even if the relationship remains unclear at first.




Comments
10 Comments
Calvin H
Just another way for pharma to limit liability while selling their poisons.
Amber Armstrong
I hear where you are coming from but it feels a bit cynical to just dismiss the whole system like that.
Many people genuinely rely on these distinctions to stay calm during treatment plans.
Fear takes over when we do not understand the terminology used by our doctors honestly.
It is easier to panic when you read death and life-threatening listed next to common cold symptoms.
We all want our patients to feel safe and protected from harmful substances definitely.
The regulations exist to catch patterns that individual doctors might miss entirely otherwise.
If nobody reported minor events how would we know about the hidden risks lurking nearby.
Community input helps update warnings faster than any internal review could possibly manage alone.
My neighbor went through a trial recently and she learned this saved her mental health significantly.
Understanding severity versus seriousness stops the endless worry loop from spinning out of control constantly.
Hospitals admit people for monitoring even if the condition itself resolves quickly within hours.
It is important to remember that mild dehydration requiring admission still counts as a serious event officially.
That nuance is exactly what prevents unnecessary alarm bells ringing for every stomach ache imaginable.
Medical records become clearer when both the patient and provider use the same vocabulary together consistently.
Reporting to MedWatch keeps the database populated with real world usage data effectively.
Even small reports help scientists spot trends that lab tests cannot detect in isolation easily.
We should encourage everyone to submit forms when they experience something weird unexpectedly.
It contributes to the safety net we all benefit from living under today hopefully.
Staying informed reduces anxiety significantly when you actually know what qualifies as dangerous behavior.
Thanks for sparking this conversation even if your take was pretty negative about the topic generally.
Victor Ortiz
You are focusing on the wrong metrics regarding safety signals entirely.
Most laypeople fail to grasp the statistical significance behind the classification thresholds.
Your emotional reaction obscures the regulatory logic that actually protects public health.
The definition hinges on outcome severity rather than subjective pain perception alone.
Ignoring the legal framework makes any critique of the system fundamentally invalid.
You might believe the data is skewed but the protocol is rigid.
I bet you haven't even read the Code of Federal Regulations section referenced in the article.
This lack of technical understanding leads to poor medical decision making eventually.
Patients need to stop treating clinical documentation as casual reading material.
The distinction between grade three severity and serious classification is legally binding.
Failure to respect these distinctions puts trial validity at significant risk.
Do not expect agencies to change definitions based on public sentiment shifts.
Real safety requires precise categorization regardless of whether it comforts patients emotionally.
Cameron Redic
Good luck explaining that to a family member in the ICU waiting room then.
Your technical precision means nothing if the patient dies from untreated confusion.
Analyst types usually overlook the human cost of bureaucracy completely.
I am sure your spreadsheet looks perfect though obviously.
Stop pretending to know better than frontline medical staff on the ground.
Dan Stoof
This information is super helpful and incredibly empowering for everyone!!
It shines a bright light on the dark corners of medical jargon!!!
Knowledge is power and this is pure enlightenment stuff!!
We can feel confident navigating trials now!!!
The clarity brought to severity scales is amazing!!!
Thank you for sharing this gem with the community!!!
RONALD FOWLER
i agree with the optimism it helps reduce stress.
the definitions are clear enough for us to understand.
staying calm is good for recovery rates.
Carolyn Kask
Foreign governments would never dare be this transparent about their safety failures ever.
Our FDA sets the gold standard for global pharmaceutical regulation compliance.
Other countries hide adverse events under mounds of red tape intentionally.
We proudly publish this data while others bury it in obscurity forever.
American innovation demands such rigorous standards for public protection.
Rest assured we are doing the best job possible for our citizens always.
William Rhodes
Your patriotism does not change the fact that safety protocols require universal application globally.
Philosophy dictates that truth transcends borders and national pride fails to cure diseases.
Being aggressive about superiority does not improve the actual chemistry involved.
We must focus on the science rather than waving flags at regulators everywhere.
Patient welfare should remain the central focus of this discussion always.
Stop using nationalism as a shield against valid criticism of systemic issues.
The goal is survival not political victory over rival agencies.
Biraju Shah
We need to enforce stricter adherence to reporting guidelines moving forward.
Patients must understand their rights and obligations clearly before signing anything.
Silence helps nobody in the safety reporting ecosystem whatsoever.
Clear communication channels between sponsors and participants are mandatory.
Disregard for these rules endangers the integrity of future research projects.
Everyone needs to prioritize accuracy over convenience in these matters.
Marwood Construction
The structural implementation of such guidelines requires careful oversight mechanisms.
Data integrity relies on consistent adherence to established reporting timelines.
Compliance ensures that aggregate statistics reflect genuine safety profiles accurately.
Professional standards dictate that all stakeholders maintain vigilance continuously.
Administrative processes must align with ethical requirements strictly.
Efficiency in reporting directly correlates with timely intervention strategies.
We must prioritize methodological rigor in all safety assessments conducted.
Write a comment